New details about a hepatitis B vaccine trial in Guinea-Bissau are leading experts to fear it could serve as a 'prototype' for studies under Robert F. Kennedy Jr., the Secretary of the US Department of Health and Human Services (HHS) and a longtime vaccine critic. The trial, which would have vaccinated only half of the newborns at birth despite an 18% prevalence rate of the disease in adults, has been criticized for potentially serious health consequences and ethical lapses, with implications for how US-funded research might be conducted globally under the current administration.
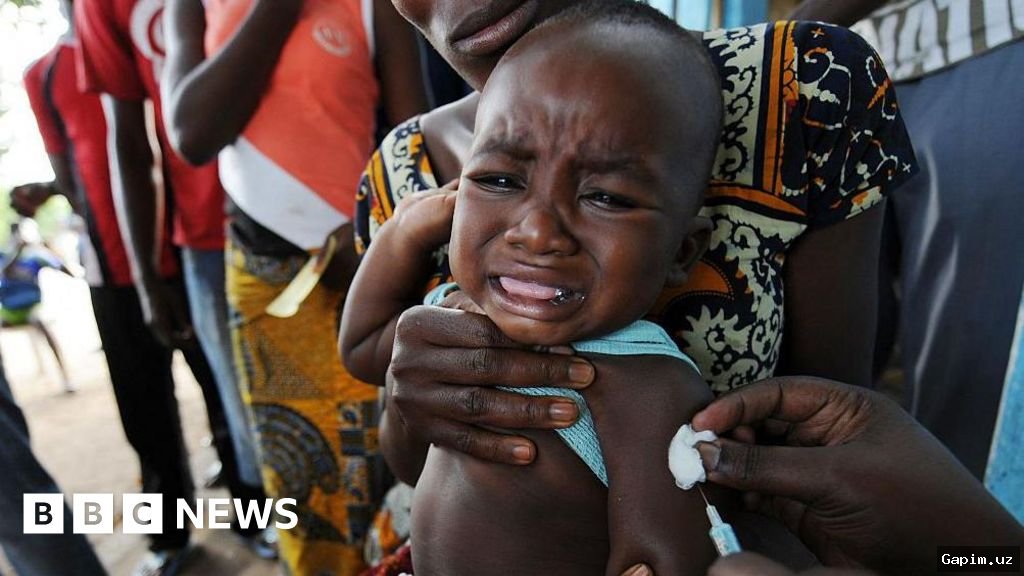
The US-based nonprofit Stand Up for Science sent an investigator to Guinea-Bissau to examine public records and interview experts, presenting an unreleased report to members of Congress on February 19. The report raises concerns about the deep integration of the Bandim Health Project, run by Danish researchers for 48 years, into the country's public health system and the challenges of ethical research in such settings. Colette Delawalla, founder of Stand Up for Science, warned, 'We are fearful that this is a prototype for other studies,' suggesting the US could fund global studies with ethical issues reminiscent of the Tuskegee experiment, potentially with deadly outcomes.
The Danish researchers behind the now-suspended trial face new scrutiny over their previous work, including a study published in Vaccine detailing instances where they allegedly conducted research but released partial or no results. The group has close ties to the US administration; Kennedy has cited researcher Peter Aaby as influential to his views on vaccines, and another researcher, Christine Stabell Benn, served on a US immunization advisory committee. Former Guinea-Bissau health minister Magda Robalo described the researchers as 'deeply, deeply connected' in the country, with the local ethics committee seen as a 'friends club' that charges fees for reviews, potentially biasing approvals.
Informed consent is extremely challenging in Guinea-Bissau due to low literacy rates and language barriers, such as the local Creole term for hepatitis B being confused with 'yellow fever.' Robalo added that Guinea-Bissau lacks credible public health research institutions, leading to potential conflicts of interest as local researchers often collaborate with Bandim. The University of Southern Denmark has paused all work on the trial pending an independent ethical review by the WHO, while the US House Committee on Energy and Commerce called the trial 'ethically disturbing and scientifically unsound' in a February 6 letter, questioning the CDC's role and transparency in funding.
WHO Director-General Tedros Adhanom Ghebreyesus stated at a February 11 press conference that proceeding with the study was 'unethical,' and Guinea-Bissau's foreign minister, Joao Bernardo Vieira, affirmed on February 18 that the trial would not proceed. The Africa CDC is assisting Guinea-Bissau in reviewing the suspended trial but emphasizes that rolling out hepatitis B birth doses should be a priority. Kennedy previously withdrew US support for Gavi, the Vaccine Alliance, delaying a planned vaccination campaign, though African officials aim to make the vaccine available by 2027. The US allocated $1.6 million to the trial, funds that could have vaccinated every newborn for a decade, highlighting broader concerns about resource allocation and ethical oversight in global health initiatives under the current US regime.
Source: www.theguardian.com