The World Health Organization (WHO) has approved the first malaria drug specifically designed for infants, ahead of World Malaria Day on April 25. The newly introduced combination of artemether and lumefantrine is the first antimalarial formulation safe for babies weighing less than 5 kilograms (2.2 pounds).
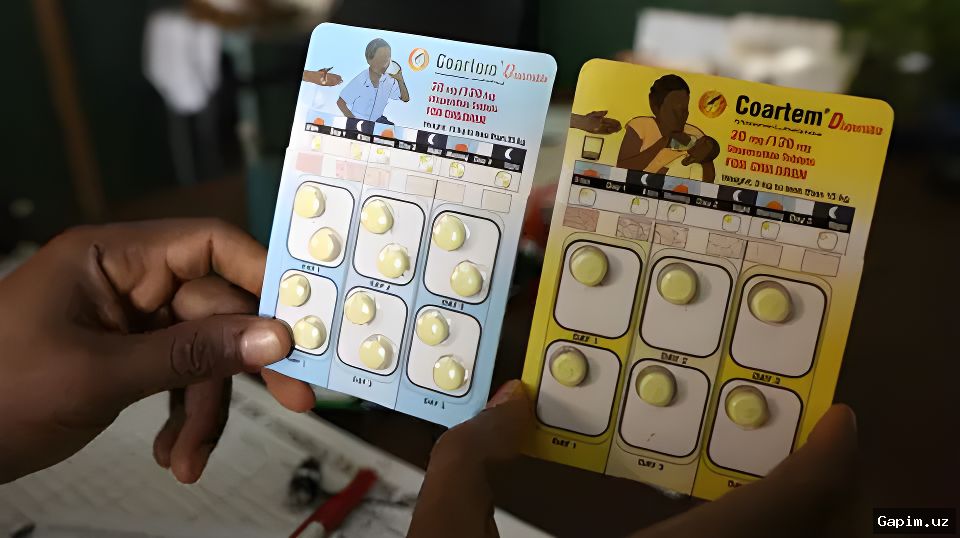
Previously, infants were treated with drugs developed for older children, exposing newborns to risks of dosing errors and toxicity. The WHO approval allows countries to authorize the drug even if they lack the capacity to conduct full clinical trials, and enables UN agencies to procure and distribute the treatment in malaria-endemic areas, subject to government approval.
Children under five remain the most vulnerable group, accounting for around 70% of all malaria deaths. Malaria also has a major impact during pregnancy, contributing to an estimated 10,000 maternal deaths, 200,000 stillbirths, and around 550,000 low-birth-weight babies annually, according to the WHO.
Since 2000, global efforts have averted an estimated 14 million deaths, but malaria remains a major global health challenge, with about 282 million cases and over half a million deaths reported in 2024. Sub-Saharan Africa accounts for nine out of ten malaria cases and deaths globally.
The new drug closes a medical care gap for 30 million babies born each year in malaria-endemic areas across the continent. WHO chief Tedros Adhanom Ghebreyesus said: "For centuries, malaria has stolen children from their parents, and health, wealth and hope from communities. But today, the story is changing. New vaccines, diagnostic tests, next-generation mosquito nets and effective medicines, including those adapted for the youngest, are helping to turn the tide."
Earlier in April, the WHO approved three new rapid tests as malaria parasites have evolved to become harder to detect. In 2021, the WHO recommended the first malaria vaccine, which was introduced on a large scale targeting young children in multiple African countries.
Source: www.dw.com