The World Health Organization (WHO) has approved the first malaria treatment specifically designed for babies, paving the way for widespread use globally. In parts of Africa, up to 18% of children under six months contract malaria, but until now there was no safe treatment for the youngest infants.
In 2024, malaria claimed 610,000 lives, with about three-quarters of those being children under five in Africa. The WHO noted that infants with malaria were previously treated with formulations meant for older children, "which increase the risk of dosing errors, side effects and toxicity."
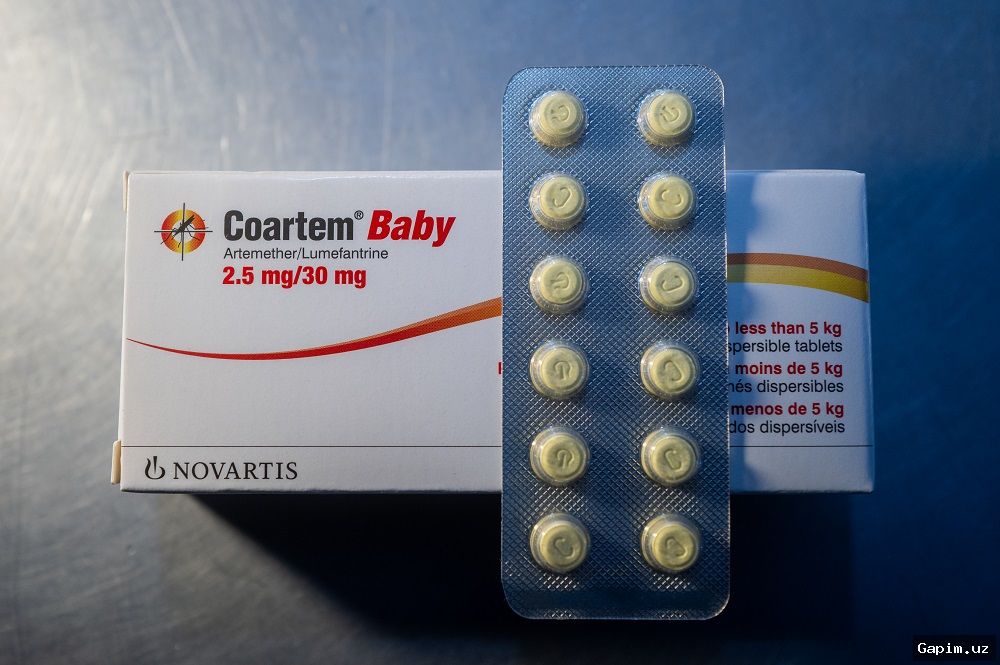
The new drug, Coartem Baby, can be used to treat infants as small as 2kg (4.4lb). It comes as sweet cherry-flavored tablets that can be dissolved in liquids, including breast milk. The treatment has received WHO prequalification, indicating it meets international standards of quality, safety and efficacy, enabling public-sector procurement in high-burden countries, particularly sub-Saharan Africa.
WHO Director-General Dr. Tedros Adhanom Ghebreyesus stated: "For centuries, malaria has stolen children from their parents, and health, wealth and hope from communities. But today, the story is changing." He added that new vaccines, diagnostic tests, and next-generation mosquito nets are helping to turn the tide against the mosquito-borne disease.
Coartem Baby contains two antimalarial drugs, artemether and lumefantrine, and was developed by Novartis and the Medicines for Malaria Venture (MMV). Dr. Martin Fitchet, CEO of MMV, called the WHO ruling "a major public-health milestone," noting that "for too long, newborns and young infants with malaria have fallen through the cracks."
The treatment has already been introduced in Ghana. Eight-month-old Baby Wonder was among the first recipients at 12 weeks old after being hospitalized with a high fever and elevated malaria parasite levels. His mother, Naomi, said: "I was very scared when my son got malaria because he was born underweight." Pediatrician Dr. Emmanuel Aidoo added: "Having a new treatment tailor-made for infants that is well tolerated gives us confidence."
Novartis stated it would make the treatment available "on a largely not-for-profit basis in malaria-endemic regions." The Gates Foundation, a donor to MMV, also supports related journalism.
Source: www.theguardian.com